Lucent Lesions of Bone
<-Axial Arthritis | Sclerotic Lesions of Bone->
Where, oh where does one start in the workup of this type of lesion? In my opinion, the first order of business is to learn the names of all of the tumors and tumor-like processes that involve bone. Richard Moser, former registrar of the AFIP, has said that among the thousands of radiology residents he taught there, there was one major cause for residents blowing unknown tumor cases: they didn’t know the names of all of the tumors. This makes sense. If you have never heard of a non-ossifying fibroma, it is unlikely that you will ever put it in your differential diagnosis. If you would cast out the demon, you must first know its name. If you don’t know Rumplestiltskin’s name…. So get busy and first learn at least the names of all of these tumors. I know what a glutealgia it is to learn all of these names. When I was a first-year resident, I thought it was ludicrous to have to do this. To me, it was as if someone had taken a bunch of histological prefixes, shaken them up in a bag, and then drawn out a few at random, stuck them together, and then added “-oma” to it to get most of the types of bone tumors. How else do you explain entities like hemangiopericytoma, angiomyolipoma, chondromyxoid fibroma, or ossifying non-ossifying fibroma? Sooooooo…. go ahead and learn the names of all of these entities. Yes, Right Now! I’ll be waiting right here when you get back.
Back already? OK! Now that you know all of the names of the tumors, let’s focus on the ones you’re likely to see in a solitary lucent expansile lesion of bone. A widely used mnemonic for this is: FEGNOMASHIC. However, I find it a lot easier to remember a mnemonic if it actually forms a real word. As it turns out, one can rearrange the letters of FEGNOMASHIC to form: FOGMACHINES. Take your choice — either works just the same. The entities considered in this mnemonic are:
Mnemonic = FOGMACHINES
Differential Diagnosis of Solitary Lucent Bone Lesions
- Fibrous Dysplasia
- Osteoblastoma
- Giant Cell Tumor
- Metastasis / Myeloma
- Aneurysmal Bone Cyst
- Chondroblastoma / Chondromyxoid Fibroma
- Hyperparathyroidism (brown tumors) / Hemangioma
- Infection
- Non-ossifying Fibroma
- Eosinophilic Granuloma / Enchondroma
- Solitary Bone Cyst
This is a fairly long differential diagnosis. However, it is one that you must learn. I still run through it every time I see one of these lesions, just to make sure that I consider all of the important possibilities.
The discussion that follows will dwell almost totally on the plain radiographic findings of these lesions. CT and MRI are wonderful tools for tumor workups, but they are fairly non-specific. Their place in the workup is to tell us where the lesion is: what its extent is; whether there are any metastases (either in the same bone or elsewhere); and whether an adjacent joint, nerve or blood vessel is involved. However, to tell us what a lesion is, the plain radiograph is still supreme. We’ve been looking at the darned things for almost a century now, and the plain film findings of most bone tumors are fairly well known. Plain films are not terribly sensitive, but they do have a decent specificity. Therefore, any workup of a bone tumor should start with a good set of plain films.
Age
Patient age is a very important bit of knowledge to have in the workup of a tumor. According to Edeiken, about 80% of malignant tumors can be correctly diagnosed on the basis of age alone. From a study of the age prevalence of 4,000 malignant bone tumors, he gives the following table:
Age vs. Malignant Tumor Type
| AGE (years) | TUMOR |
| 1 | neuroblastoma |
| 1 – 10 | Ewing’s of tubular bones |
| 10 – 30 | osteosarcoma, Ewing’s of flat bones |
| 30 – 40 | reticulum cell sarcoma (Primary histiocytic lymphoma), fibrosarcoma, parosteal osteosarcoma, malignant giant cell tumor, lymphoma |
| 40 + | metastatic carcinoma, multiple myeloma, chondrosarcoma |
Size
What does the size of a lesion tell us? Unfortunately, not a whole lot about the histological type of lesion that we are dealing with. However, describing the size of a lesion is part of another important aspect of tumor management: pretreatment staging of the extent of the lesion.
Surgery is the preferred treatment for many lesions in this category. The surgeon wants to know where to cut so as to remove all of the lesion, along with an area of normal tissue on all margins of the tumor. Lesion size as measured on plain films will not tell us this accurately — if anything, it tends to greatly underestimate the extent of the lesion. However, it provides a convenient first-order approximation of the extent (a lower bound on its size) while the early workup is being arranged. Definitive assessment of the pretreatment extent of a lesion will require MR or CT.
Margins
This is one of the most important things that you can determine about a solitary, lucent, expansile lesion of bone. Why is this? Because, this is the finding that will give you your best shot at determining the biological activity of the lesion (how fast is it growing?). This is important, because in general, the faster a process grows, the more likely it is to be malignant.
So, how can we determine biological activity from the margin of the lesion? The answer is bone response. Bone is sensitive to a variety of stimuli, and generally responds to one of the processes in FOGMACHINES by either removing bone or creating bone. That’s right! The bone itself does the removing or creating of bone – not the disorder involving the bone. At the AFIP, they are fond of saying that the only things that can remove bone are osteoclasts and orthopedic surgeons. I agree with this rule, but would also add talented amateurs to the list (lawnmower and saw accidents, auto crashes, blast injuries, and animal bites are favored mechanisms of bone removal by amateurs — professionals prefer saws, drills, and osteotomes and confine their efforts to operating rooms). For this reason, the term “expansile lesion” is a bit of a misnomer, since the lesion itself is not expanding the bone. The bone is remodeling itself in response to the stimulus of the lesion.
The other thing to know about bone response is that while it is certain, it is rather slow. If a lesion is growing slowly, then the bone will have plenty of time to retreat from the lesion, removing some bone around the lesion, but also laying down new bone around the margins of the lesion. This generally has the effect of producing a sclerotic and usually distinct margin around the lesion. If process grows more rapidly, the bone may only have time to retreat before the lesion, and not have time to lay down this sclerotic rim. Solitary lucent lesions in bone with a distinct margin are generally called “geographic” lesions, whether or not they have a sclerotic rim.
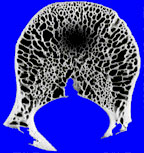 |
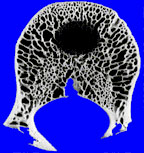 |
| geographic lesion with ill-defined rim | geographic lesion with well-defined rim |
If the lesion grows more rapidly still, there may not be time for the bone to retreat in an orderly manner, and the margin may become ill-defined. Rather than a single discrete lesion, we may see several ill-defined foci of lucency. This has been termed a “moth-eaten” pattern.
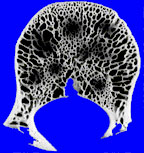
a “moth-eaten” lesion
If, alas, the process grows more rapidly still, then the bone’s retreat may become disorderly indeed. Continuing this battlefield analogy, the boundary between normal and abnormal bone may be lost altogether, with only a very ill-defined pattern of lucency seen, caused by many small, irregular holes in the bone, left behind by osteoclasts. This is an extremely aggressive pattern, sometimes called a “permeative” pattern.
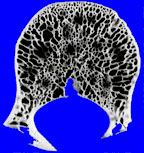 |
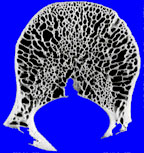 |
| permeative pattern | normal bone |
The presence of a permeative pattern usually means that the patient either has an aggressive infection or a malignant tumor. The most common malignancies that give this pattern are metastases, myeloma, primary histiocytic lymphoma, and Ewing’s sarcoma. These lesions are sometimes referred to as “round cell lesions” due to the small, dark, round cells that they display to the pathologist.
Matrix
What is matrix, anyway? It is stuff produced by osteoblasts and chondroblasts that eventually becomes, respectively, normal bone and cartilage. Bone tumors form matrix just as a normal bone does, but sometimes in greater quantity. Also, matrix produced by tumors is usually quite abnormal, and does not ossify properly. Why do we look for tumor matrix? Because, it helps us to give a bone tumor a rough histological classification into one of three categories: cartilage-producing, bone-producing or other. Cartilaginous tumors (enchondroma, chondrosarcoma, chondromyxoid fibroma, etc.) will tend to produce cartilaginous matrix, while tumors from the osteoid series (osteoma, osteoblastoma, osteosarcoma, etc.) will tend to produce osseous matrix. In order to see matrix on plain radiographs, it has to calcify. Chondroid matrix, for example tends to produce small punctate or swirled areas of calcification. Adjectives applied to this cartilaginous matrix include “popcorn-like”, “curvilinear”, or “speckled”. Osseous matrix tends to be dense and confluent, and invokes descriptive terms like “cloud-like” or “mashed potatoes”. Other lesions tend to produce little or no calcification in their matrix (fibrous dysplasia, fibrosarcoma, malignant fibrous histiocytoma, solitary bone cyst, etc.). Although the term “ground-glass” has been applied to this appearance of matrix, I think that it is a bit confusing, since a fogged film with no diagnostic information on it has a ground-glass appearance also. If I don’t see any definite calcified matrix in a lesion, I prefer to just say that instead.
Location
Most expansile, lucent lesions are located in the medullary space of the bone. However, we can further define the location of the lesion by noting its relationship to the physis. Many lesions tend to occur in a “favorite” part of the bone. The favored locations are listed in the figure below.
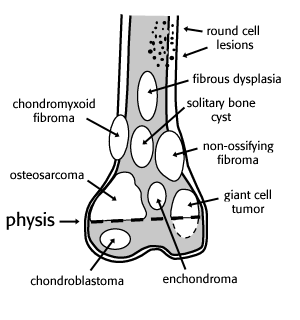
figure after Madewell, et al 1981
Epiphysis
Very few lesions tend to arise in the epiphysis. Chondroblastoma is one of the very few tumors that arise here before the physis closes. Osteomyelitis can also arise in the epiphysis.
Several entities can spread across the physis. Osteomyelitis is a classic example of this. Although the dogma for years has been that malignancies such as osteosarcoma and Ewing’s tumor rarely cross the physis, more recent experience with MRI has shown this to be untrue. With very sensitive pulse sequences such as STIR (short-tau inversion recovery), subtle extension across the physeal plate may be seen not uncommonly.
After the plate closes, the physis ceases to be an anatomic barrier to disease, and a variety of lesions can be seen involving the epiphyseal area, such as giant cell tumor, enchondroma or aneurysmal bone cyst. Helpful tips in steering the differential diagnosis among these entities include the facts that most enchondromas will exhibit chondroid matrix, most giant cell tumors will abut an articular margin, and most aneurysmal bone cysts appear, well, “aneurysmal” or expansile. It was once thought that aggressive tumors, such as Ewing’s tumor and osteosarcoma tended to “respect” the physeal plate and only rarely cross it. However, more recent studies (Panuel,Norton) of the behavior of such tumors with sensitive MR pulse sequences show that osteosarcomas may cross the plate into the epiphysis in 70 – 80 % of cases and Ewing’s tumor in about 20 %.
Metaphysis
This is the fastest growing area of a bone, and also the most likely area for a primary neoplasm to arise. This is especially true in the distal femur and proximal tibia, which are the fastest growing metaphyseal areas in the skeleton. The metaphysis also has the best blood supply of the bone, so entities such as infection or metastasis will commonly be seen in this area as well. In general, most of the entities in FOGMACHINES (with the exception of chondroblastoma) will be most commonly seen in the metaphyseal area of a given bone.
Diaphysis
Most of the entities in FOGMACHINES can also appear in the diaphysis, although with less frequency. Notable exceptions are: chondroblastoma, which almost always occurs in an epiphysis or epiphyseal equivalent (most apophyses, the patella and the calcaneus); giant cell tumor, which almost always occurs in an apophysis or in the bone adjacent to a join space; osteoblastoma, which usually occurs in the posterior elements of the spine; and aneurysmal bone cyst, which is usually metaphyseal in location.
Periosteal Reaction
Periosteal reaction is an important finding to note in the workup of bone tumors. However, it also occurs due to several other processes besides tumors. Therefore, I have given periosteal reaction a chapter all to itself. Please refer to this chapter on periosteal reaction for a discussion of how it relates to bone tumors.
Multiplicity
So, what do you do if the patient has multiple lucent lesions? Well, you go through pretty much the same thought processes that you went through for a solitary lesion. The main thing that is different is the differential diagnosis that you use. A lot of the entities in FOGMACHINES don’t really make sense as a cause of multiple lucent lesions. However, some of them do. It turns out that one can simply trim out the entities that don’t make sense and what’s left works just fine for multiple lesions. Thus, our differential reduces like this:
Differential Diagnosis of Multiple Lucent Bone Lesions
Mnemonic = FOGMACHINES –> FEMHI
- Fibrous Dysplasia
- Metastasis / Myeloma
- Hyperparathyroidism (brown tumors) / Hemangioma
- Infection
- Eosinophilic Granuloma / Enchondroma
This leaves the letters FMHIE as our differential for multiple lucent lesions. Try as I might, I still haven’t been able to come up with any kind of decent word out of these letters. Clyde Helms orders them as FEMHI, and you can make up your own order if you like. If you do come up with a real word in English or some other language out of these five letters, tell me what it is and I will buy you a taco.
Wise Sayings About Solitary Lucent Lesions
Sometimes, all the logical principles that you have at your disposal don’t seem to help very much, and one must fall back on some of the empirical maxims that musculoskeletal radiologists have accumulated over the years. Here are a few of the ones I have used over the years.
- With a long lesion in a long bone, think of fibrous dysplasia.
- Simple cyst, enchondroma, and fibrous dysplasia can mimic each other and can be hard to distinguish. Thus, when you think of one of these three entities, also think of the other two.
- Giant cell tumors nearly always occur near a joint surface.
- Certain bones in the body can be considered “epiphyseal equivalents” for purposes of differential diagnosis. These include the patella, the calcaneus, and most apophyses. Therefore, for lucent lesions in these areas, one should include the classic epiphyseal entities such as chondroblastoma, giant cell tumors and aneurysmal bone cysts.
- Lucent lesions of the sternum should be considered malignant until proven otherwise (Helms CA, personal communication, 1983).
- Keep in mind that the classic descriptions of bone tumors that you spend so much time studying are for untreated lesions. What kind of lesions do radiologists spend most of their time looking at? Treated lesions — treated with surgery, chemotherapy, cryotherapy and radiation therapy. In surgically treated lesions, besides simple resection of the lesion, one may also see replacement by a metal prosthesis, an allograft, or other forms of bone grafting. In short, you won’t see the “classic” appearance of a lesion for very long in a given patient. When the patient first presents to you, you may not even have any history of these prior interventions, so you will just have to remember this phenomenon. Also, any given film that you see of a patient is just one frame out of a long documentary movie about that patient — movies change. Remember this.
- Edeiken J. Roentgen diagnosis of diseases of bone. (4th ed.) Baltimore: Williams & Wilkins, 1989 (Harris JJ, ed. Golden’s diagnostic radiology).
- Madewell JE, Ragsdale BD, Sweet DE. Radiologic and pathologic analysis of solitary bone lesions. Part I: Internal margins.Radiol Clin N Am 1981;19:715-748.
- Panuel M, Gentet JC, Scheiner C, Jouve JL, Bollini G, Petit P, Bourliere-Najean B, Devred P. Physeal and epiphyseal extent of primary malignant bone tumors in childhood. Correlation of preoperative MRI and the pathologic examination. Pediatr Radiol 1993;23:421-424.
- Norton KI, Hermann G, Abdelwahab IF, Klein MJ, Granowetter LF, Rabinowitz JG. Epiphyseal involvement in osteosarcoma.Radiology 1991;180:813-816.